Detoxicare Clinical Trials
"Trust Detoxicare's seasoned management team with over 20 years of expertise to guide you through your trials with confidence."

ABOUT DETOXICARE
Detoxicare Molecular Diagnostics Laboratory is a 100% Filipino-owned and operated molecular laboratory in the Philippines.
Established in 2019, our mission is to serve the Filipino people with diagnostics tests that will help them make better and wiser decisions for their healthcare.
PCR Test, or polymerase chain reaction, is a molecular diagnostic test used for detecting various pathogens, from bacteria, tuberculosis, and viruses including SARS-CoV-2, the virus that causes COVID-19.
Thus, Detoxicare Molecular Diagnostics Laboratory is among the first private RT-PCR test laboratories in Philippines accredited by the Department of Health to test for COVID-19.
Beyond being a DOH accredited COVID-19 testing provider, we offer molecular diagnostics for the detection of various respiratory diseases and the genetic activity of cancer tumors.
THE CLINICAL TRIAL CENTER
Detoxicare Clinical Trial Center, newly established in Mandaluyong City, Metro Manila, Philippines. Strategically positioned, the Center is 2 kilometers from Mandaluyong City Medical Center, 5 kilometers from Ortigas Center in Pasig City, the nearest business district, and 12 kilometers from the airport.
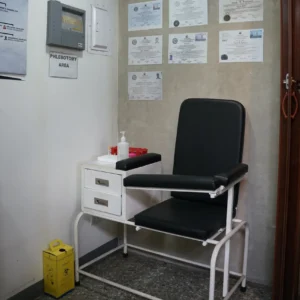
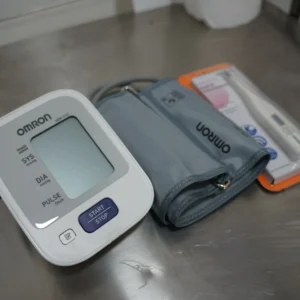
CLINICAL TRIAL CENTER FACILITIES
The Center is fully equipped for most Phase II to IV out-patient trials
OTHER SERVICES
- Mobile X-Ray
- Mobile ECG
- Monitoring Area
- Storage Area for Paper Documents
- 24-hour Power Supply
- WiFi
CLINICAL TRIAL CENTER FACILITIES
The Center is fully equipped for most Phase II to IV out-patient trials
DETOXICARE LABORATORY
Detoxicare utilizes cutting-edge technologies in molecular diagnostics, offering a broad range of advanced testing services. The laboratory is equipped with capabilities for immunohistochemistry (IHC), fluorescence in situ hybridization (FISH), polymerase chain reaction (PCR), and next-generation sequencing (NGS). These sophisticated tools enable highly accurate and sensitive diagnostics. Detoxicare’s primary portfolio includes comprehensive testing for the diagnosis of infectious diseases and the identification of critical oncology biomarkers, supporting precision medicine and personalized treatment strategies.
MANAGEMENT TEAM
The Clinical Trial Center is managed by an expert team with more than 20 years of experience from Harmony Trials OPC, a newly established Site Management Organization.
Rodmar C. Pulido
 Since 2000, Rodmar has been an active figure in the clinical research industry in the Philippines, having roles in monitoring, operations management, and business development. He founded a clinical research center at one of the Philippines’ premier hospitals and was the inaugural General Manager of a start-up CRO for one of the country’s largest conglomerates. In his operations role, Rodmar has been instrumental in overseeing various facets including monitoring, data management, statistics, medical writing, quality assurance, and training throughout the Philippines. His business development efforts have extended across Southeast Asia, Japan, South Korea, the European Union, and the USA. Rodmar’s achievements include pioneering the introduction of biosimilar and gene-therapy trials in the Philippines and launching the nation’s first comprehensive data management, statistics, and medical writing services for a Pan-Asian CRO. An active member of the Philippine Clinical Research Professionals Inc., he plays a key role in engaging stakeholders and delivering GCP training to diverse institutions. Rodmar also served as the organization’s President in 2019 and consulted on the operational framework for the National Clinical Trial & Translational Center of the Philippine Government in 2024.
Since 2000, Rodmar has been an active figure in the clinical research industry in the Philippines, having roles in monitoring, operations management, and business development. He founded a clinical research center at one of the Philippines’ premier hospitals and was the inaugural General Manager of a start-up CRO for one of the country’s largest conglomerates. In his operations role, Rodmar has been instrumental in overseeing various facets including monitoring, data management, statistics, medical writing, quality assurance, and training throughout the Philippines. His business development efforts have extended across Southeast Asia, Japan, South Korea, the European Union, and the USA. Rodmar’s achievements include pioneering the introduction of biosimilar and gene-therapy trials in the Philippines and launching the nation’s first comprehensive data management, statistics, and medical writing services for a Pan-Asian CRO. An active member of the Philippine Clinical Research Professionals Inc., he plays a key role in engaging stakeholders and delivering GCP training to diverse institutions. Rodmar also served as the organization’s President in 2019 and consulted on the operational framework for the National Clinical Trial & Translational Center of the Philippine Government in 2024.
Carina O. Barlongo
 Carina is a dedicated clinical research professional and licensed Medical Technologist with over 25 years of experience in clinical trials. She specializes in Phase III-IV and post-marketing surveillance studies. Her career began at Novartis Philippines, where she served as a Clinical Research Associate, implementing both local and global trials. Her exemplary performance led to her promotion as Clinical Operations Manager, a role she held until 2017, overseeing the entire clinical trial operations. Throughout her career, Carina has managed trials across various therapeutic areas, including Cardiology and Metabolism, Dermatology, Gastroenterology, Oncology, Ophthalmology, Psychiatry, Respiratory, and Rheumatology. More recently, she has contributed significantly to COVID-19 vaccine trials.
Carina is a dedicated clinical research professional and licensed Medical Technologist with over 25 years of experience in clinical trials. She specializes in Phase III-IV and post-marketing surveillance studies. Her career began at Novartis Philippines, where she served as a Clinical Research Associate, implementing both local and global trials. Her exemplary performance led to her promotion as Clinical Operations Manager, a role she held until 2017, overseeing the entire clinical trial operations. Throughout her career, Carina has managed trials across various therapeutic areas, including Cardiology and Metabolism, Dermatology, Gastroenterology, Oncology, Ophthalmology, Psychiatry, Respiratory, and Rheumatology. More recently, she has contributed significantly to COVID-19 vaccine trials.
Marivic P. Pangilinan
 Marivic is a licensed medical technologist with extensive experience in clinical research and project management for almost 20 years. She started her career as a research assistant at the Department of Health Promotion and Education at the University of the Philippines Manila College of Public Health and then as a university researcher at the Department of Physiology at the University of the Philippines Manila College of Medicine. The Year 2000 was when she started her clinical research profession at AstraZeneca Pharmaceuticals Philippines, Inc., where, during her tenure, she advanced from Clinical Research Associate to Clinical Project Manager. She has managed Phase III-IV global studies on therapeutic areas including respiratory and cardiovascular diseases, CNS, oncology, infectious diseases, diabetes, and vaccination. Among her significant achievements are successful inspections by the Philippine FDA and Japan FDA. She was honored with the AstraZeneca CRR-Asia Pacific Quality Award (2005–2006) and the Regional Medical Director’s Award (2003). Recently, she has significantly contributed to the COVID-19 vaccine trials as one of the project leads.
Marivic is a licensed medical technologist with extensive experience in clinical research and project management for almost 20 years. She started her career as a research assistant at the Department of Health Promotion and Education at the University of the Philippines Manila College of Public Health and then as a university researcher at the Department of Physiology at the University of the Philippines Manila College of Medicine. The Year 2000 was when she started her clinical research profession at AstraZeneca Pharmaceuticals Philippines, Inc., where, during her tenure, she advanced from Clinical Research Associate to Clinical Project Manager. She has managed Phase III-IV global studies on therapeutic areas including respiratory and cardiovascular diseases, CNS, oncology, infectious diseases, diabetes, and vaccination. Among her significant achievements are successful inspections by the Philippine FDA and Japan FDA. She was honored with the AstraZeneca CRR-Asia Pacific Quality Award (2005–2006) and the Regional Medical Director’s Award (2003). Recently, she has significantly contributed to the COVID-19 vaccine trials as one of the project leads.
Kimberly Joy R. Semon
 Kim is an experienced medical laboratory professional with 34 years of clinical experience both locally (various laboratories in major hospitals) and internationally (Labcorp, USA). She specializes in molecular diagnostics, transplant, and personalized medicine. She has 15 years of focused experience in cellular therapy practice and trials, including the first Philippine Government funded stem-cell trial. Additionally, she has co-authored several publications in the field of cellular therapy. Kim is a licensed Medical Technologist and holds a master’s degree in business administration with major in Health.
Kim is an experienced medical laboratory professional with 34 years of clinical experience both locally (various laboratories in major hospitals) and internationally (Labcorp, USA). She specializes in molecular diagnostics, transplant, and personalized medicine. She has 15 years of focused experience in cellular therapy practice and trials, including the first Philippine Government funded stem-cell trial. Additionally, she has co-authored several publications in the field of cellular therapy. Kim is a licensed Medical Technologist and holds a master’s degree in business administration with major in Health.
INVESTIGATORS
Harold Henrison Chiu, RCh, MD
Doctor of Medicine: University of the Philippines Manila
Residency Training in Internal Medicine: Philippine General Hospital
Fellowship Training in Endocrinology, Diabetes & Metabolism: Philippine General Hospital
Clinical & Research Fellowship: Diabetes Center, Taichung Veterans General Hospital, Taiwan ROC
Interests:
Diabetology
Diet & Nutrition
Endocrinology
Internal Medicine
Transgender Health and Wellness
Maria Nikki Cruz, MD, FPCP, DPCEDM
Doctor of Medicine: University of Santo Tomas
Residency Training in Internal Medicine: St. Luke’s Medical Center
Fellowship Training in Endocrinology, Diabetes & Metabolism: St. Luke’s Medical Center
Interests:
Diabetology
Endocrinology
General Medicine
Internal Medicine
Jane Suaco, MD, FPCP, FPSMID
Doctor of Medicine: St. Luke’s Medical Center College of Medicine
Residency Training in Internal Medicine: St. Luke’s Medical Center
Fellowship Training in Infectious Diseases and Tropical Medicine: St. Luke’s Medical Center
Interests:
COVID-19
Dengue
Tuberculosis
Pneumonia
Urinary Tract Infections
Skin Infections
Infectious Disease
Tropical Disease
Internal Medicine
Jamie Lynn Tiu Co, MD, FPCP, DPCCP
Doctor of Medicine: University of the Philippines Manila
Residency Training in Internal Medicine: Cardinal Santos Medical Center
Fellowship Training in Pulmonary Medicine: St. Luke’s Medical Center
Interests:
General Medicine
Internal Medicine
Pulmonology
THE COMMUNITY
Centrally located within Metro Manila, Mandaluyong City is surrounded by Manila, San Juan, Makati, and Pasig. Covering approximately 9 square kilometers, it boasts a resident population of around half a million, swelling to 1.2 million during the day. With a population density of about 46,000 per square kilometer and a youthful median age of 21 years, Mandaluyong City thrives at the heart of urban dynamism.
The chart highlights that five of the top ten leading causes of illness are respiratory-related, ranging from respiratory tract infections to pneumonia. Additionally, prevalent illnesses include skin diseases, urinary tract infections, cardiovascular diseases, and diabetes among others. Notably, Upper Respiratory Tract Infection consistently reported the highest number of cases per 100,000 population in 2015, 2016, and 2017, with Hypertension, Diabetes, and UTIs also showing persistent prevalence.
Morbidity Count and Ten Most Common Causes of Illness: 2017
Cause of Illness
No Data Found
We collaborate with the leaders of the city and its component districts or barangays, and 20 district health centers to effectively recruit participants
References: Philippine Statistics Authority, City of Mandaluyong websites
2020 Census of Population and Housing Results (City of Mandaluyong) | Philippine Statistics Authority | National Capital Region (psa.gov.ph)
Social Services and Status of Well-being – City of Mandaluyong
WE’RE HERE TO HELP
If you have any questions about us, or if you would like more information about how you can participate or collaborate with us, please don’t hesitate to get in touch. We are committed to providing you with the best support and information.
Other Contacts
Office
DETOXICARE MOLECULAR DIGANOSTICS LABORATORY
Highpointe Medical Hub, 241 Shaw Blvd,
Mandaluyong, Metro Manila, Philippines
Get Directions >